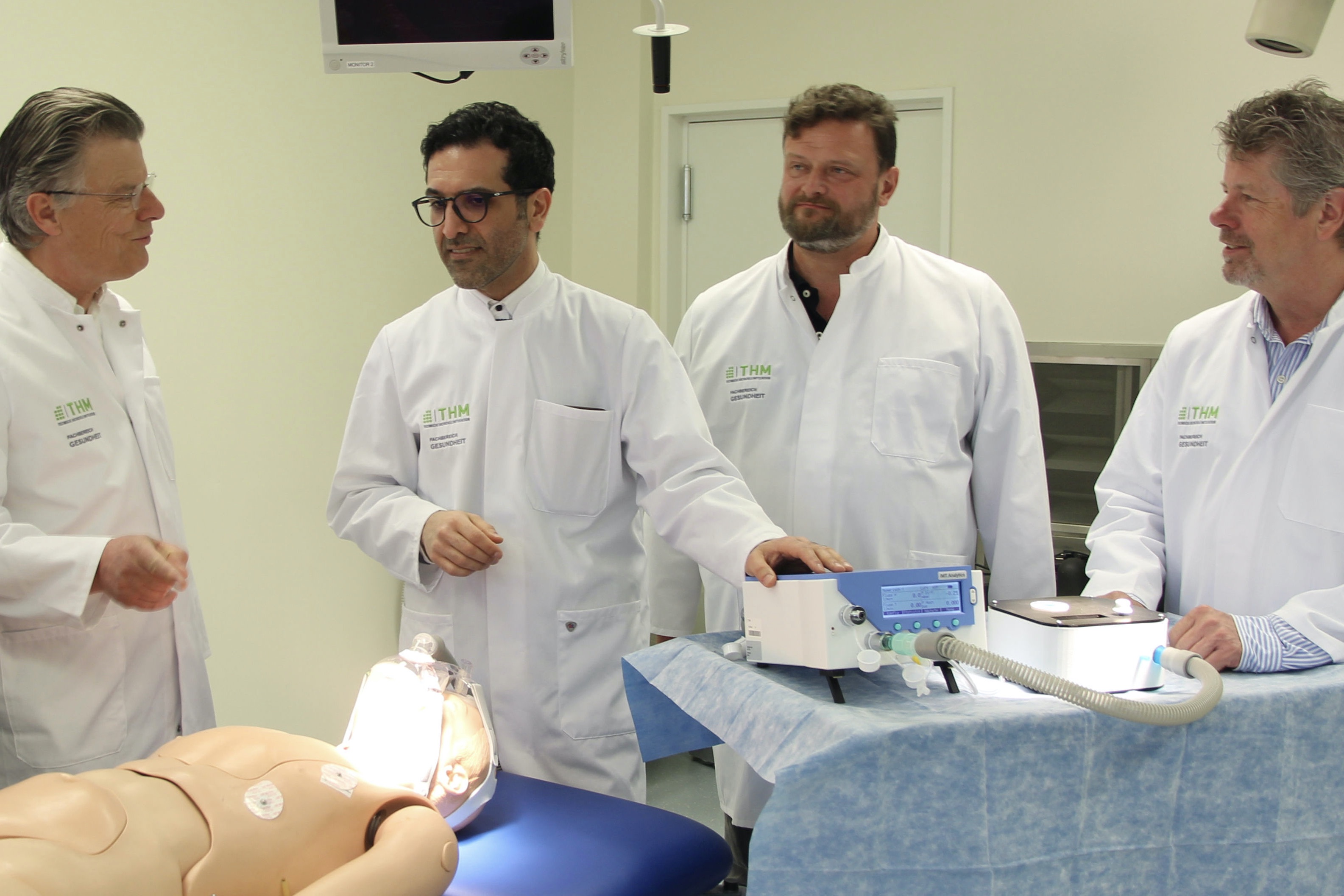
Respirators are now a scarce commodity. Scientists from the Technical University of Central Hesse (THM) and the University Hospital Giessen/Marburg (UKGM) want to change this: They have developed a simple and cost-effective solution for the ventilation of Covid-19 patients: the “Mobile Single Ventilation Device 2020.”
If severe pneumonia occurs in Covid-19 patients, pronounced respiratory distress occurs, requiring invasive ventilation. But ventilators are limited. Professors Dr. Volker Gross and Dr. Keywan Sohrabi, from the THM’s Department of Health, and Prof. Dr. Ulrich Koehler, head of the Sleep Medicine Centre at the UKGM, are developing a solution. Their mobile single ventilator 2020 (MEB 2020-1) is based, among other things, on findings from sleep medicine. “The system actively supports patients in breathing,” explains Dr. Koehler. “It thus protects the patient’s body and maintains the vital concentration of oxygen in the blood. Patients receive an increased pressure during inhalation, which is lowered during exhalation. This procedure is known in specialist circles as Biphasic Positive Airway Pressure. The positive pressure during exhalation prevents the collapse of the airways. The device is also used to administer oxygen.”
Ventilator can also be operated by non-experts
The device is technically simple and mobile. “Our system can be used before and after invasive ventilation. The respiratory support is non-invasive,” adds Dr. Sohrabi. “Today, it is performed with expensive multifunctional devices. MEB 2020-1 generates sufficient inspiratory pressure and ensures that the small airways do not collapse during exhalation. During development, we also took care to avoid air contamination. Many current concepts neglect this aspect.” And the MEB 2020-1 should also be usable by non-specialists, for example by the patients themselves, relatives or carers. This ease-of-use will take the burden off highly qualified specialist staff. The device also dispenses with complicated monitoring technology. Nevertheless, it works reliably and in accordance with the applicable medical-grade monitoring standards. Currently, the scientists are testing a prototype; with the support of the Giessen-based medical technology company MHM, the scientists are also preparing for production.