Biopharmaceuticals are often unstable as liquids and therefore have poor shelf life. This includes their raw materials, such as peptides and olionucleides, which are necessary for the production of important biopharmaceutical drugs. Freeze-drying is one way to bring these sensitive molecules into a stable form. Optima pharma GmbH in Gladenbach, Germany develops and produces these special systems, which are also suitable for these active pharmaceutical ingredients.

Frozen, pressurized, heated: This sounds like the steps of some sort of painful procedure. In fact, they are actually the daily routine in the manufacturing process for biopharmaceuticals and many other drugs, providing a gentle method to remove excess water. The result is the desired active ingredient in the form of a finely powdered substance.
Today freeze-drying, known technically as lyophilization, is a method of choice for the pharmaceutical industry. This process very gently converts antibiotics, hormones, antibodies, RNA-based vaccines, and many other medicines into a storable, solid powder form. “While patients usually receive these medicines in liquid form rather than as tablets, as a liquid formulation the usually sensitive molecules are not particularly stable. They can change in structure and thus lose their effectiveness. In addition, there is a risk of germs in the water,” explains Stephan Reuter, Managing Director of Optima Pharma GmbH at the Gladenbach site in central Hessen. The company specializes in the development and production of systems for freeze-drying.
Frozen water gently evaporated
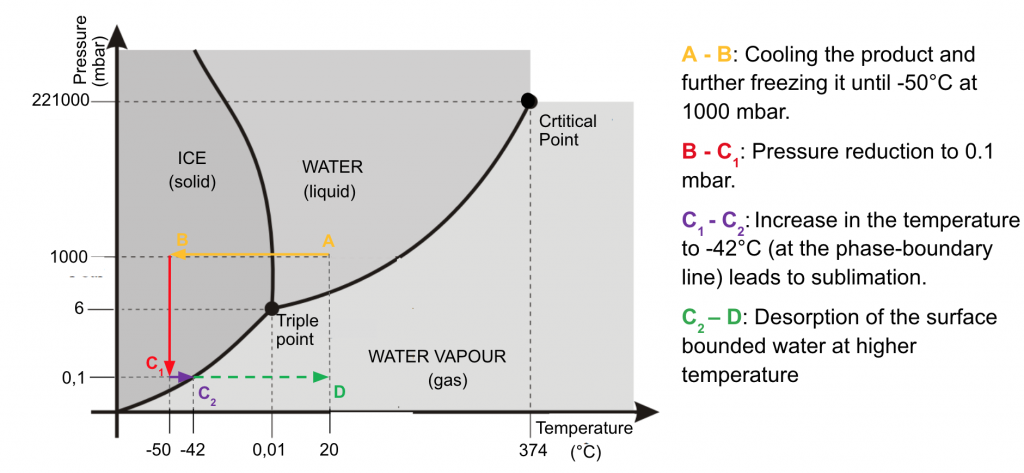
But what makes freeze-drying a gentle process? A key aspect is the frozen water that evaporates under vacuum (sublimation) while the temperature is gently raised again. Ideally, only the desired product remains, and this is in the form of a fine powder. Another way of extracting a dissolved substance from water is familiar to anyone who cooks: boiling until liquid water has evaporated entirely. However, this latter approach is anything but gentle. The great danger is that the product will be irreversibly damaged by sustained high temperatures. In addition, it is not only water that evaporates during heating; the desired product can also change to a gaseous state and is thus lost. “Freeze-drying, on the other hand, is a safe method and time-tested for converting even sensitive biopharmaceuticals into a stable and storable product,” Reuter says.
Biopharmaceuticals: freeze-drying is preferred
Due to factors like the Corona pandemic, as well as the growing number of biological pharmaceuticals and their transport over long distances, demand for freeze-drying continues to increase. Optima Pharma has doubled its production area in Gladenbach, where their freeze-drying equipment is manufactured, and the number of employees at the site has also almost doubled since 2014. The company’s export share is more than 85 percent. With an average growth rate of 7-10 percent, the company can look optimistically to the future. Reuter, himself, is confident that this positive trend will continue. “Already, about one-fifth of the top 100 pharmaceuticals are freeze-dried, and for biologics it’s already almost half,” Reuter says. As an engineer, Reuter brings two decades of professional and management experience in the pharmaceutical industry to the work. He foresees methods for continuous product development. “The demand for this method has also increased the requirements for the freeze-drying process itself. As a result, we develop innovative, customized solutions for these needs.”
Peptides as promising drug candidates
The freeze-drying of peptides in particular requires complex production processes and special knowledge. Peptides are small proteins, i.e. protein molecules. They consist of several amino acids linked by peptide bonds. Peptides have numerous functions – some act as messenger substances in plants, for example, and others as hormones in the human body. Due to their properties, there are numerous potential applications for these small proteins in molecular biology, immunology and (bio)medicine. For cancer patients, for example, peptides offer the chance for individualized vaccines to be produced in the future – and thus a promising strategy for effective therapy. “Peptides are highly effective, and they exhibit high selectivity and specificity with regard to their biological targets,” Reuter explains. “Peptides thus offer a promising prospect for novel drug designs and are of great advantage for drug development.”

The fine art of freeze-drying
Because of their great versatility, freeze-drying equipment must also be designed to meet the needs of peptide production, the final stage of which involves solid-liquid separation. “In this process, the lyophilisate, also known as freeze-drying cake, is obtained at our customer’s facility in containers that resemble baking trays,” Reuter says. The final stable peptides are then available from milligrams to kilograms in a fine powder form and are filled into vials or glass tubes. “The fact is, there is no off-the-shelf freeze dryer that works. We have to adapt the equipment to the particular pharmaceutical product, the customer’s wishes and the circumstances on site. This is our special know-how as a special equipment manufacturer,” Reuter says. The company is also involved in a promising area of development: controlled nucleation. The focus here is on the freezing process, which is difficult to control, especially for high-purity products. Uneven freezing has a corresponding effect on the ice structure. However, controlled nucleation is a process that solves this problem very successfully: Additional pressure is built up in the system, which is released at an accelerated rate. This results in the formation of extremely homogeneous ice crystals. At the same time, drying is accelerated.
Turning on energy-saving
Shorter process times in turn reduce the use of consumables – and, above all, energy. “Despite the great advantages that freeze-drying offers, it is, unfortunately, also a very energy-intensive process, and correspondingly costly,” explains Reuter. “After all, the steel plants, which can weigh up to 30 tons, must first be sterilized with hot steam, cooled to freezing temperatures of up to minus 70 degrees Celsius, placed under vacuum and then reheated to up to 130 degrees Celsius. This is the only way to achieve the desired temperatures inside,” Reuter adds. “As a result, drug manufacturers are often reluctant to consider the technology for their products.” However, refrigeration technology, using new high-performance refrigerants, yields significant savings: Using a dedicated refrigeration system lowers energy consumption. Further, new innovative refrigerants also save 10 to 20 percent in energy. Taking into account the savings from shorter process times, in total freeze drying plants today consume up to a quarter less energy than before these measures were implemented.
Focus on medical trends
Optima Pharma is also working to establish alternative refrigerants and make its technologies more sustainable and environmentally friendly. The reason: Conventional refrigerants unfortunately harbor a high greenhouse potential. The company’s other development goals arise due to trends in the pharmaceutical industry, for example, personalized medicine, mRNA and gene and cell technologies. “These changes influence the production facilities of drug manufacturers, which require fewer large plants, but rather produce decentrally and with smaller equipment,” Reuter says. “Analogous to batch sizes, biopharmaceutical drug projects tend to install smaller freeze dryers, and often multiple freeze dryers in the same move. Even hospital pharmacies are potentially interested in our systems.” As a result, the footprint per freeze dryer or the number of units is a key factor.
Today’s freeze-drying process has become much more energy-efficient and more diverse than it was just a few years ago. Optima Pharma has made it its business to keep an eye on industry trends and to further develop process technologies accordingly – thus making an important contribution to innovative, high-quality pharmaceuticals.