The search for a vaccine to contain the current coronavirus pandemic is in full swing. A vaccine that was originally developed against the coronavirus MERS, a disease related to SARS-CoV-2, is now the focus of a Marburg research team.
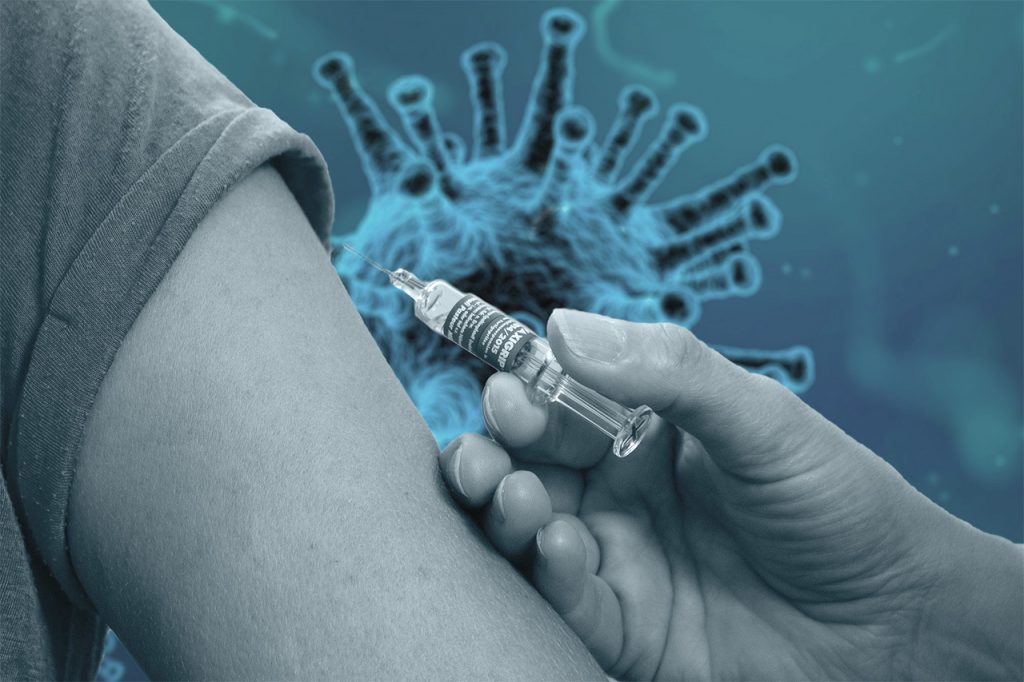
The key to winning the battle against the SARS coronavirus 2 is an effective vaccine. Researchers all over the world are looking for the right active substance, including medical experts in Central Hessen. There, a team of scientists from the Philipps University of Marburg – along with colleagues from the Ludwig Maximilian University of Munich and the University Medical Center Hamburg-Eppendorf (UKE) – have now come close to a possible solution: The use of the experimental MERS agent MVA-MERS-S to help against SARS-CoV-2.
In cooperation with the German Centre for Infection Research (DZIF), this MERS vaccine is currently being tested for tolerance and efficacy against COVID-19 in a clinical study involving 23 volunteers. Professor Stephan Becker, head of the Institute of Virology at the Philipps University of Marburg and coordinator for Emerging Infectious Diseases at DZIF, is investigating the first immune response of the antibodies from his laboratory’s study participants. “These results show that the new vaccine has the potential to be used in possible future MERS outbreaks,” he says.
SARS and MERS viruses: similar in structure
MERS-CoV viruses are the cause of a serious respiratory disease, Middle East Respiratory Syndrome (MERS). This respiratory disease is fatal in up to 35 percent of cases. To date, more than 2,500 cases of MERS have been confirmed in 27 countries worldwide. Since 2012, sporadic outbreaks have occurred repeatedly in Arab countries. Like SARS-CoV-2, MERS-CoV is a type of coronavirus. Since the surface proteins of the virus’ outer envelope, so-called spike proteins, are similar in structure, they can serve as key proteins for an antiviral agent.
MVA-MERS-S as the basis for a vaccine
If Becker’s clinical tests are successful, the MVA-MERS-S vaccine may serve as the basis for a SARS-CoV-2 vaccine. “The development of the MERS vaccine forms the basis on which we at DZIF are rapidly developing a vaccine against the new virus,” adds Prof. Marylyn Addo, Head of the Infectiology Section at the UKE in Hamburg.
The researchers are using a kind of modular principle for this purpose. The vaccine candidate MVA-MERS-S is based on an attenuated virus (Modified Ankara Virus – MVA) that has already been used to eradicate smallpox. Using genetic engineering methods, the MVA virus can theoretically be supplemented with protein components of the SARS-CoV-2 coronavirus. Thus, the vaccination “feigns” an infection with SARS-CoV-2 in the human immune system, which stimulates an immune response. The body should now trigger the formation of specific antibodies and T-cells against the alleged Covid-19 disease and build up a vaccination protection that can also ward off a real infection.
Only clinical tests guarantee effectiveness
Since it sounds so straightforward to develop this vaccine, the public may wonder if it will actually take until 2021 before this SARS-CoV-2 vaccine is ready. The issue here is clinical vaccination studies in humans: These involve several lengthy phases. It must be guaranteed that the upcoming vaccine is safe and well tolerated. It must also be clarified how often and at what intervals the vaccine should be administered.
Currently, the World Health Organization (WHO) counts about 70 vaccine projects against Covid-19 (as of April 2020). It would be advantageous if as many vaccines as possible could be approved and produced using many production facilities. In the meanwhile, Professor Stephan Becker and his team from Middle Hessen are contributing to the worldwide fight against coronavirus.


I cannot find a date on your article. It would be great to know when this (and other articles) are written.
I would thank you very much for doing so!
Marijke, Devon AB Canada
Dear Marijke, this article was posted in May 2020.